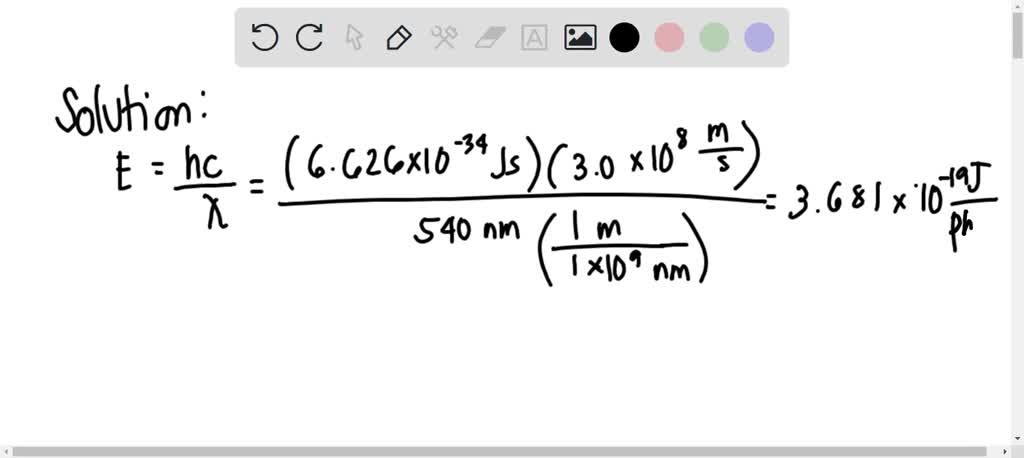
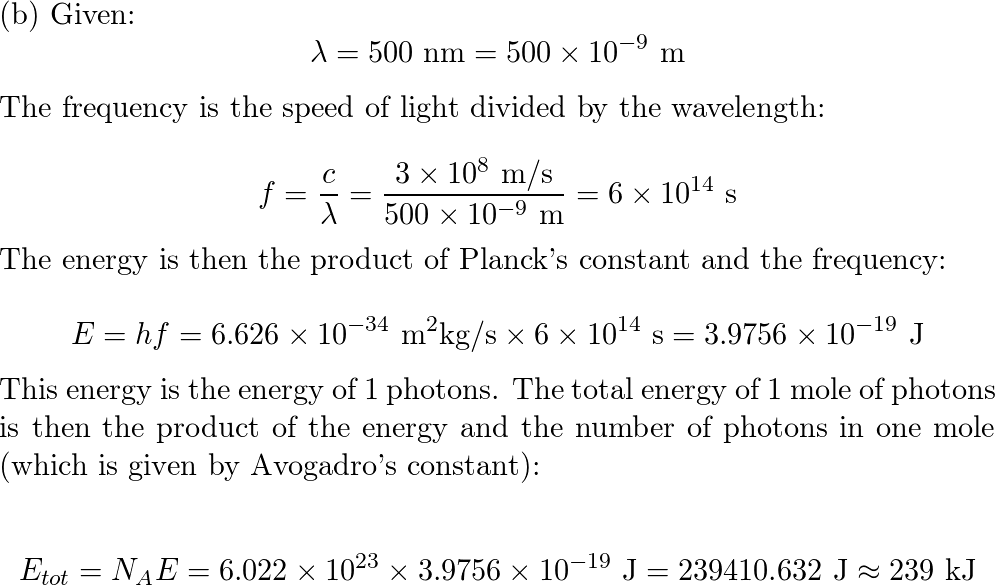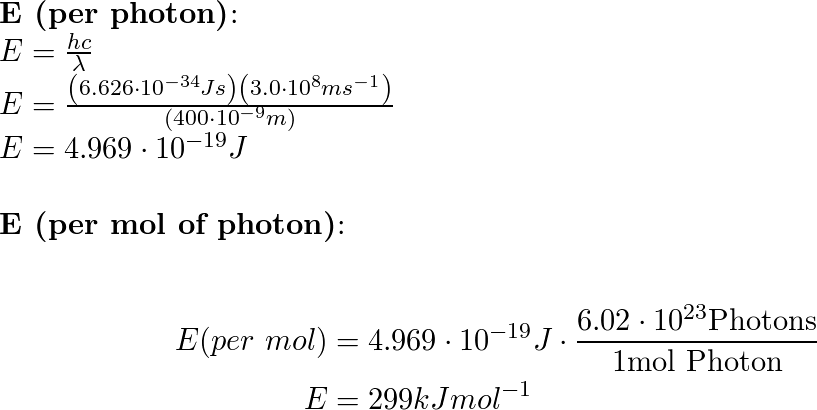calculate energy of one mole of photons of radiation whose frequency is `5xx10^(14) hz`... - YouTube

Calculate the energy of one mole of photons of radiation whose frequency is `5 xx 10^(14) Hz`. - YouTube
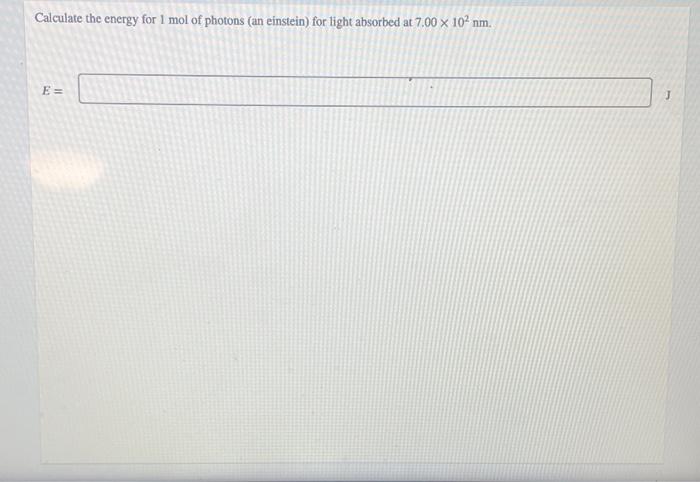
Welcome to Chem Zipper.com......: Calculate the energy per mole of photon of electromagnetic radiations of wavelength 4000Å.

The Gibbs energy change accompanying a given process is - 85.77 kJ mol ^-1 at 25 ^o C and - 83.68 kJ mol ^-1 at 35 ^o C. The heat of reaction of process at 30 ^o C is: