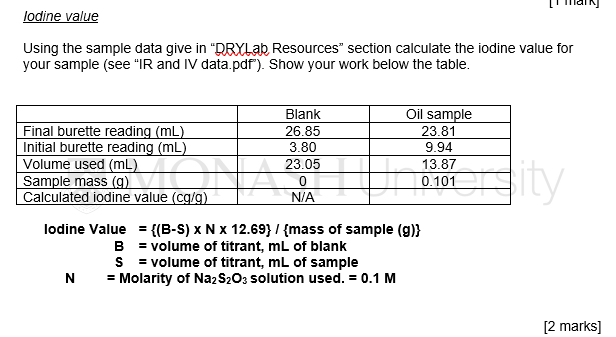
Rapid Iodine Value Estimation Using a Handheld Raman Spectrometer for On-Site, Reagent-Free Authentication of Edible Oils | ACS Omega

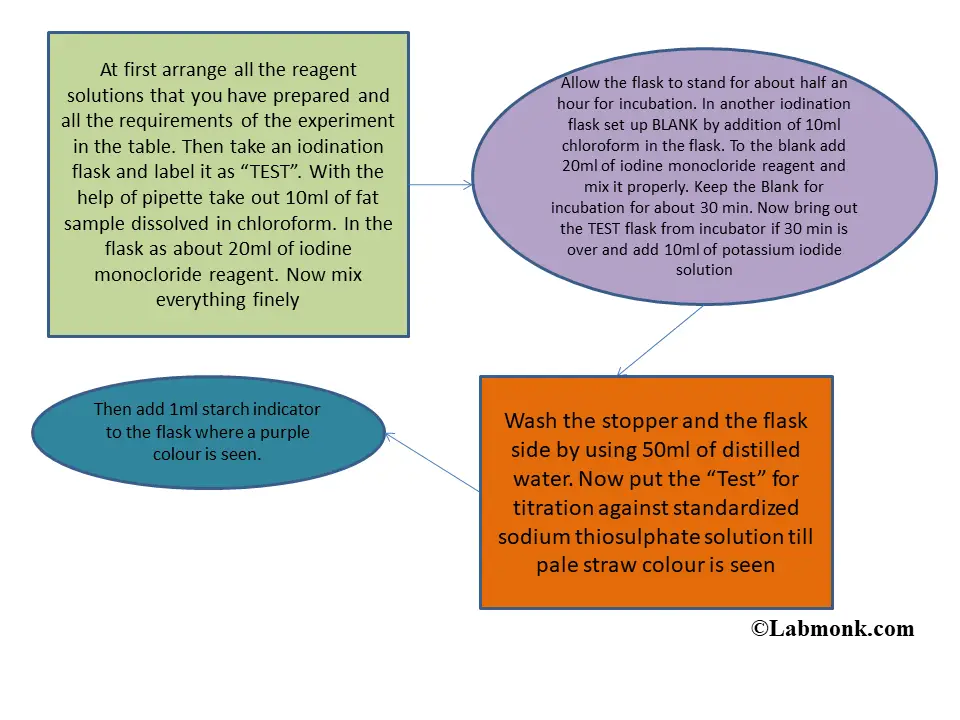
Iodine number, Saponification number, Acid Number, Reichert-Meissl (RM) number & Rancidity of Oil/Fats | The Pharmapedia
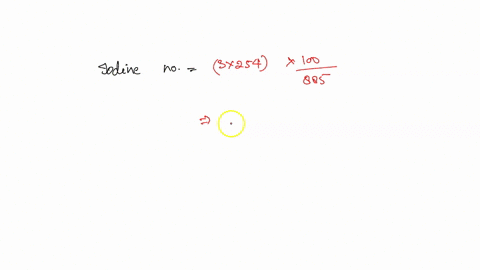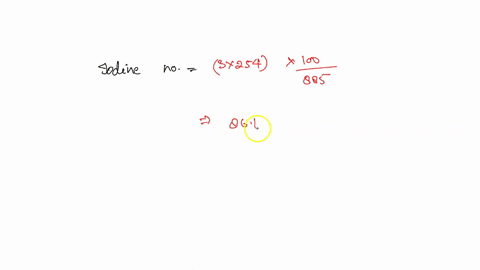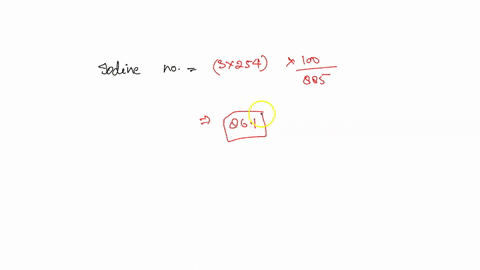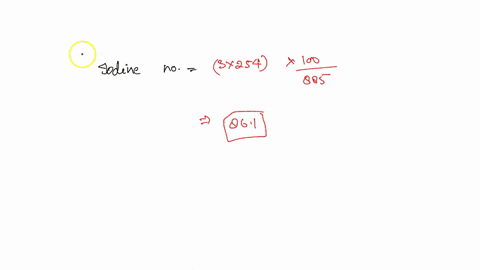
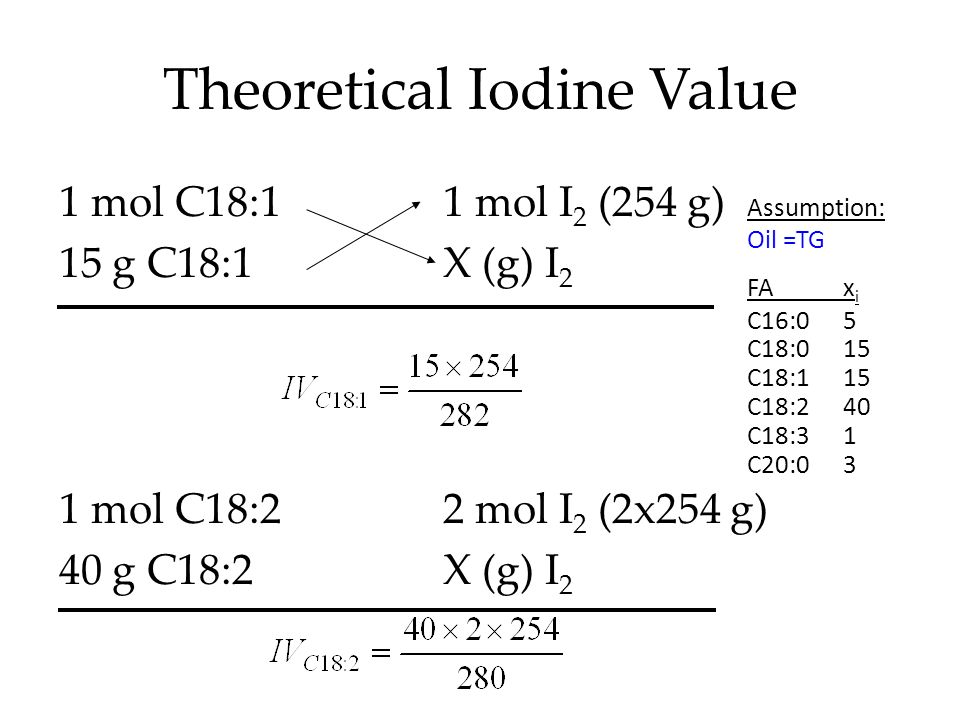
SOLVED: how do i calculate the theoretical Iodine number of triolein with molar mass as 885 g/mol and the molar mass of iodine is 254 g/mol.

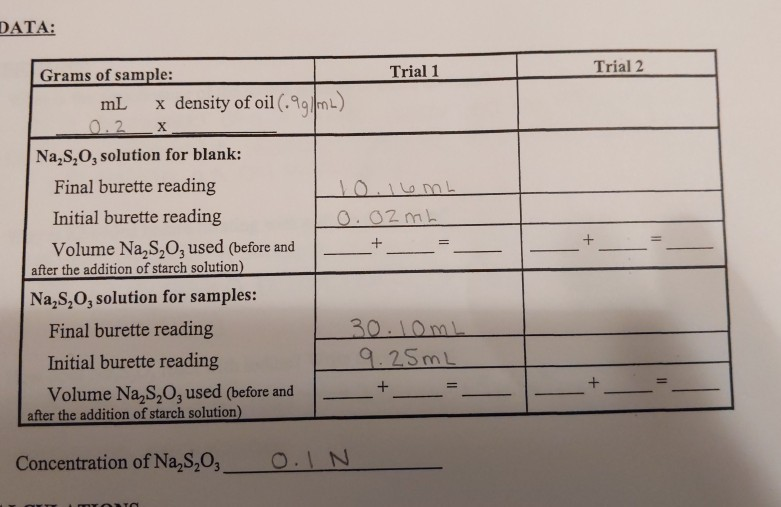
SOLVED: LIPid LAB DATA AND REPORT FORM Hanus Method for Determination of lodine Number Unknown YOUR DATA: WEIGHT OF OIL SAMPLE: 0.350 VOLUME OF TITRANT USED FOR TITRATION: 40.OmL CALCULATE IODINE NUMBER: